Ensuring Global Compliance With:
Core Capabilities of Octalsoft BMS
End-to-End Biosample Lifecycle Management
- Centralized biorepository database software for collection, storage, retrieval, and disposal.
- Barcode/RFID-enabled sample tracking with real-time location mapping.
- Automates chain-of-custody with complete sample audit trails.
Compliance-Ready for Regulated Environments
- Designed to meet FDA, HIPAA, GDPR, and GxP standards.
- Audit-ready reporting for inspections and certifications.
- Ensures full compliance for clinical biorepository management systems.
Advanced Data Management & Analysis
- Built-in biorepository data management tools for reporting and trend analysis.
- AI-driven insights to monitor storage conditions, usage trends, and inventory levels.
- Connects biosample metadata with laboratory data management software.
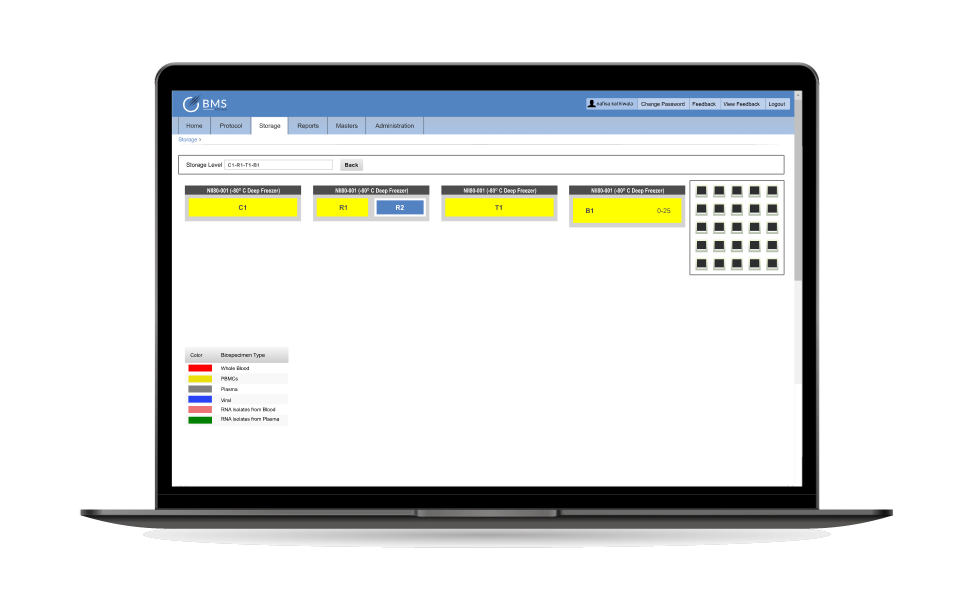
Integrated Biosample Storage & Logistics
- Temperature and condition monitoring for sensitive biological samples.
- Manages freezers, cryogenic storage, and specialized containers.
- Enables bi-directional integration with LIMS laboratory software and clinical databases.
Scalable for Pharma, Biotech, and Clinical Research
- Supports large-scale global repositories and collaborative networks.
- Customizable workflows for biorepository management in pharma and biotech.
- Cloud-enabled for multi-site biobank operations.
Resources That Speak
for Octalsoft BMS

eClinical Suite Case Study

Factsheet – Biorepository Management System (BMS)
Enhance Your BMS
with These Powerful Add-ons
Key Benefits for Every
Role in Your Organization
Business Leaders
Maximize ROI through efficient biosample data management and reduced sample loss. Accelerate drug development cycles with traceable, reliable biorepositories that enhance decision-making.Lab Managers & Scientists
Simplify daily operations with intuitive biorepository management software. Ensure sample accuracy and integrity through automated quality controls and real-time tracking.Clinical Research Teams
Maintain secure, compliant handling of trial biospecimens across all study phases. Seamlessly integrate with clinical LIMS and data management systems for unified oversight.Quality & Compliance Officers
Ensure continuous regulatory readiness with centralized biorepository data management. Access audit-ready logs, storage conditions, and complete sample histories anytime, anywhere.Why Choose Octalsoft?
Domain Expertise
With decades of combined experience in clinical research technology, our team understands the needs of CROs, Sponsors, and Sites.
All-in-One Platform
From planning and operations to data capture and analytics - we deliver integrated platforms for every stage of the trial lifecycle.
Regulatory Ready
Our systems are designed to meet global regulatory standards such as 21 CFR Part 11, GCP, and GDPR, ensuring compliance at every level.
24/7 Support
We offer personalized onboarding, 24/7 support, and ongoing optimization services to ensure long-term success for your studies.










 Download
Download